MSI Applications and Analytical Case Study
MSI in drug development
In drug development, it is crucial to visually monitor in-vivo pharmacokinetics and distribution of administered drugs to specific tissues and local areas to understand the characteristics of drugs. Autoradiography with radioisotopes (RI) has been conventionally used as a precise method for evaluating distribution in the body; however, it is difficult for this method to separately detect unchanged compounds and their metabolites. MSI is anticipated to serve as a complementary method to autoradiography in drug development research, as it can distinguish and detect unchanged compounds and their metabolites.
By merging with images from hematoxylin-eosin staining and immunohistochemical staining obtained through optical microscopes, MSI allows for the observation of compound localization along with morphological and pathological characteristics. This capability also enables the confirmation of the extent to which the compounds have reached their target tissues. MSI is expected to be utilized for evaluation of drug efficacy in target tissues, confirmation of Proof of Concept (POC), and clarification of mechanisms of toxicity expressions.
The types of drugs that can be analyzed using MSI include small-molecule compounds, nucleic acid medicines, and peptide drugs. Moreover, its applications are not limited to pharmaceuticals but can also be extended to a variety of samples and compounds such as food and agrochemicals, indicating a wide range of potential application fields.

Flow of MSI
We utilize Matrix Assisted Laser Desorption Ionization (MALDI) as the ionization method.

- Animal experiment
- Resection and freezing of target tissue
- Preparation of frozen section
- Spraying of matrix as an ionization assisting agent onto the frozen section slice adhered onto the glass slide dedicated to MSI
- Analysis with MSI equipment
The laser beam is focused onto a specific area at regular intervals on the section slide. There are two modes of analysis: “whole analysis” with wide intervals to understand the distribution of the whole tissue and “high resolution analysis” with narrow intervals to see the distribution in detail.
For MSI, we have equipped our facility with solariX from Bruker, which offers ultra-high resolution mass spectrometry at the world's highest level. This enables precise determination of the molecular weights of target compounds.

Case study: Evaluation of chloroquine localization in the eyeballs of pigmented rats
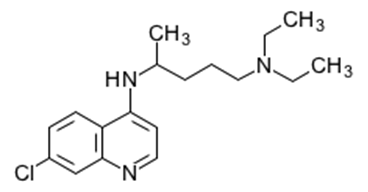
We used MSI to evaluate the localization of chloroquine in rat eyeballs.
Previous studies have shown that chloroquine administered to rats accumulates in the uvea (retina) of the eyeball, based on results from whole-body autoradiography. Additionally, toxic effects include vacuolation of the retinal nerve fiber layer and hyperplasia of the pigmented epithelium. However, the detailed distribution of chloroquine in these regions was not previously known.
Chloroquine was administered to pigmented rats by single oral dosing (6, 20, 100 mg/kg), and the eyeballs were resected on the day after dosing. Frozen sections were prepared from the resected eyeballs, and whole analysis and high-resolution analysis of the ocular tissues were conducted by MSI.
Quantitative analysis can be conducted by analyzing chloroquine standard solutions of known concentrations.

Quantitation regions were selected in the retinal outer layer (A) and the retinal inner layer (B) of each eyeball (two regions for each layer). The means of the results are shown below.

MSI quantitative value (μg/g,mean)
| No. | A:Rerinal outer layer | B:Rerinal inner layer |
|---|---|---|
| 101 | <LLOQ | <LLOQ |
| 103 | 12.6 | <LLOQ |
| 105 | 79.8 | 4.02 |
| 107 | 271 | 43.9 |
LLOQ : 1 µg/g
In addition to analyzing the whole tissue section, high-resolution analysis of selected regions is also possible. By high-resolution analysis with laser irradiation every 15 micrometers in the yellow squared area, it was found that chloroquine was mainly localized in the pigmented epithelium and choroid outside of it.
By comparing the distribution image of chloroquine, distribution image of A2E (specific marker of pigmented epithelium on retinal cell layer), and HE stain image of the section after MSI analysis, it was confirmed chloroquine was distributed in the choroid and pigmented epithelium.